Lentiviral Vectors for Cell Therapy
Lentiviral Vectors for Cell Therapy
Supporting Lentiviral Vector Development for Safer and More Efficacious Cell Therapies
Complex lentiviral vector designs for CAR T cells are necessary for successful regulatory approved ex vivo gene therapies/cell therapies such as Kymriah (Novartis) to treat acute lymphoblastic leukemia. SIRION Biotech’s mission is to enable cell therapy developers to develop tomorrow’s cell therapies with our tools, technologies, and expertise in a field that is evolving quickly from both a scientific and a regulatory perspective.
SIRION provides a 360° end-to-end solution within the R&D and preclinical lentivirus space to develop safe and efficacious lentivirus-based drug products.
We offer capabilities to develop the four key components of a lentivirus-based drug product:
-
Lentiviral vector backbone optimized for clinical application
-
Therapeutic expression cassette development
-
Lentiviral surface modification
-
Manufacturing process tools and development
SIRION KEY Advantages
De-risking
All our projects profit from more than 12 years in-house expertise in lentivirus vector technology and the main criteria for developing LV vectors for efficacious and safe drug products.
We are constantly updating and expanding our technological portfolio according to the latest scientific insights and are also excited to develop new assets in collaboration with our CGT clients.
Integrated development
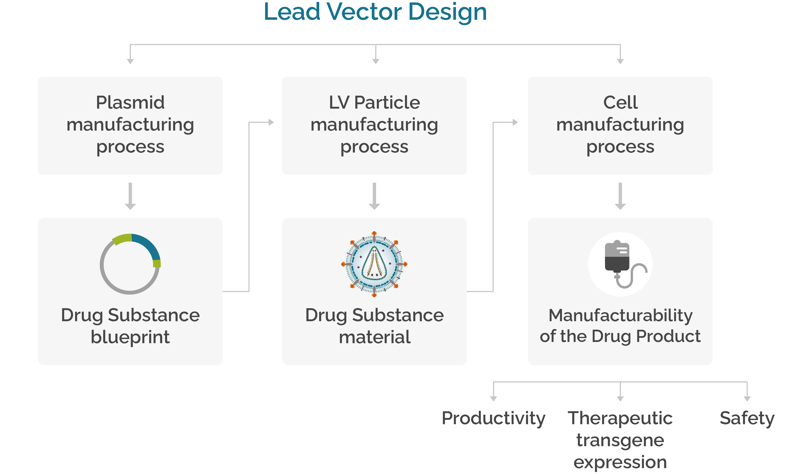
Our experts consider the vector design as the basis for a successful manufacturing of the viral particles and the cell manufacturing process. We carefully consider the deep connection between the lead vector design and any challenge encountered during the development of a cell and gene therapy, including quality, potency and cost effectiveness.
Needs and Technology Focus
Our specialists are trained to support cell therapy developers by carefully discussing and identifying our clients needs. Project feasibility as well as a portfolio of development and manufacturing options are addressed in a sophisticated project set-up.
Developing LV vectors for Efficacious and Safe Drug Products
Lead vector development
Manufacturing technologies and tech transfer
-
Optimized proprietary manufacturing processes to supply particles for research and preclinical studies of ex vivo and in vivo gene therapies.
-
In-house process development to overcome challenges with customized solutions.
-
Bioinformatic analysis of the manufacturing metrics of over 6,000 vectors produced at SIRION provide an excellent internal dataset to identify fundamental parameters for vector design and particle manufacturing.
-
Characterization and documentation of vectors, particles and manufacturing process according to the most recent regulatory guidelines serve as basic material for the GMP manufacturing of the lentiviral particles and regulatory filing.
-
Patented transduction enhancer technology for GMP manufacturing of ex vivo gene therapies.
Manufacturing for Preclinical Studies
Manufacturing of lentiviral particles for research follows our ISO certified manufacturing procedure.
Our experienced process development team also establishes advanced manufacturing procedures on demand for individual requirements for experimental validation and the experimental workflow, while addressing key clinical requirements for the lentiviral vector product – safety, efficacy, and manufacturability – as early as possible.
Our preclinical manufacturing processes – including custom developed advanced manufacturing procedures – are transferrable to partners within our closely aligned, worldwide CDMO network for GMP-manufacturing. This significantly speeds up the transfer process. We can additionally transfer the plasmid sequences for LV drug substance manufacturing and provide physical plasmid starting material via plasmid CDMO partners.
LentiBOOST Transduction Enhancer
Lead vector design and optimized lentiviral particles are just the first part of a successful lentiviral cell therapy. The cell material and manufacturing process also contribute extensively to the manufacturability and performance of the whole therapy. LentiBOOST is SIRION Biotech’s proprietary lentiviral transduction enhancer which boosts transduction rates and VCN up to 90% in a wide range of clinically relevant cell types.
LentiBOOST transduction enhancer is currently used in more than 35 Phase III and Phase I/II clinical trials worldwide as well as one approved product.
