Adenoviral Vectors for Vaccination
Adenoviral Vectors for Vaccination
Adenoviral Vectors for Vaccination
Adenoviral Vectors for Vaccination
Adenoviral vectors are a highly promising technology for application in gene therapy, as genetic vaccines, and as oncolytic viruses for treating cancer (Gao et al. 2019). SIRION offers three technology platforms for supporting therapy developers working with adenoviral vectors.
AD19A/64 Vector
SIRION Biotech’s proprietary Ad19a/64 vector (Subtype D) can be used to overcome the low transduction efficiency of commonly used Ad serotypes and help overcome the challenge of pre-existing immunity [1] It is an attractive alternative for the most commonly used human adenovirus type 5 for specific types of applications.
Ad19a/64 is the first accessible subtype D adenovirus vector. This vector provides superior transduction of human cells (e.g. dendritic cells, cardiomyocytes, fibroblasts, PBMCs, myoblasts, and myotubes) when compared to adenovirus type 5 (Ad5). It also possesses CAR independent binding properties, targeting sialic acid-dependent receptors. In preclinical vaccine studies, the vector induced superior levels of CD4 and CD8 immune responses in non-human primates.
Key Benefits of our Ad19a/64 vector
- Target new cell types/tissue due to its unique receptor affinities
- Leverage its low seroprevalence within the human population
- Superior vaccine vector platform compared to Ad5
- Overcome limitations due to CAR dependent transduction
Transduction experiments with human myoblasts and primary myotubes
Human Myoblasts
Figure 3. Transduction of myoblasts with Ad5-EGFP (Ad5) and Ad19a/64-EGFP (Ad19a). Primary myoblasts of human, ape, pig and mouse origin were transduced with 1250 VP/cell (open columns), 2500 VP/cell (shaded columns), and 12500 VP/cell (solid columns) respectively, and GFP expression was quantified after 48 hr. Each value represents the mean of transgene expression per well in relative light units (RLU) from five independent experiments +/- SD.
Human Myotubes
Figure 4. Transduction of primary myotubes (MTs) with Ad5EGFP and Ad19a/64-EGFP. Ten-day-old myotubes were transduced with 1250 VP/cell (open columns). 2500 VP/cell (shaded columns) and 12500 VP/cell (solid columns), respectively. GFP expression was quantified after 48 hr. Each value represents the mean relative light units (RLU) of five independent transductions +/- SD.
Ad19a/64 in Mucosal Boost Immunization Against SARS-CoV-2
In the course of the worldwide pandemic caused by SARS-CoV-2, German scientists tested Ad5 as well as Ad19a/64 to administer Spike S protein intranasally in mice [3]. After plasmid DNA or mRNA priming before intranasal immunization, the group saw a strong systemic and mucosal immune reaction triggered by both vectors. The authors have shown the potential of using adenoviral vectors to boost mucosal immunity in mice. Additionally, the group described slight differences in the tropism of Ad5 and Ad19a/64 that requires further investigation, especially when translating from animal studies to clinical testing.
Licensing Information
If you are interested in this technology, please get in touch with our Business Development and Licensing team at: licensing@sirion-biotech.com or use our contact form.
Publications
BAC Technology – Building Viral Vectors from Scratch

Classical cloning strategies are difficult and largely ineffective for viral vectors harboring large genomes (e.g. AV can carry transgenes up to 36kb). SIRION Biotech’s proprietary viral cloning technology offers a refined solution benefiting from the large capacity of bacterial artificial chromosomes (BAC).
The system is built in a modular way and consists of two major elements:
-
Component 1: A small shuttle vector (pO6A5) into which the desired transgene is cloned.
-
Component 2: Electrocompetent BA5-FRT E. coli cells that carry a single BAC vector conferring resistance to chloramphenicol. The BAC vector includes the replication deficient viral genome.
Following transformation of the shuttle vector into the electrocompetent BA5-FRT E. coli cells, Flp recombinase mediated recombination between the shuttle and the BAC vector occurs.
Due to a highly sophisticated selection system, only cells containing recombined BAC vector can grow and form bacterial colonies. As a result, almost 100% of screened colonies contain BAC vectors with the correctly recombined transgene. This feature greatly increases the ease of use for the cloning system as it requires very little ‘hands on’ time from shuttle vector transformation to the isolation of the virus particles. When applied to adenoviral vectors the virus yields and quality are comparable to those obtained with established adenoviral cloning systems. The technology can easily be applied for the generation of large AV expression libraries.
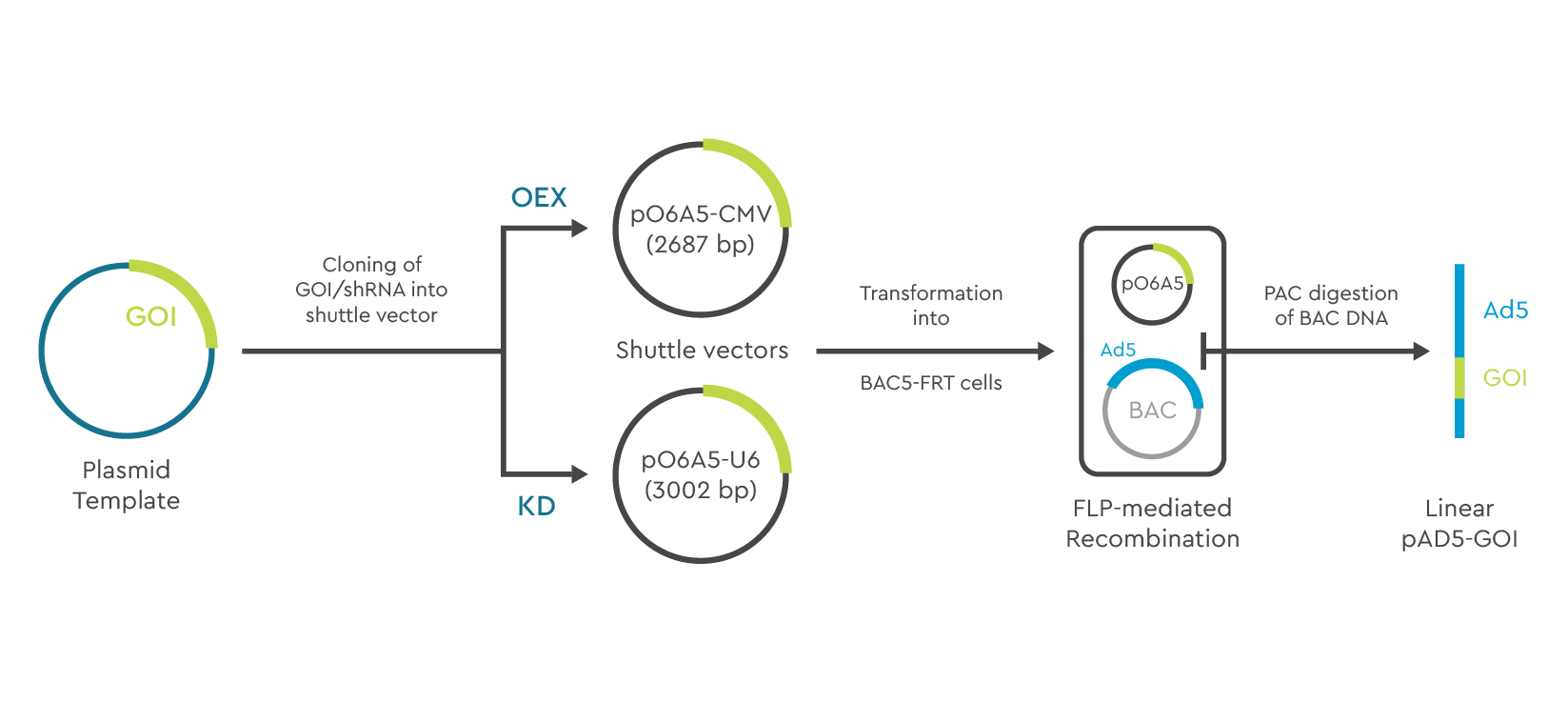
Figure 6. Example of the BAC technology using E1/E3 deficient Ad5 genome.
-
Cloning of your gene of interest (GOI) or shRNA into the pO6A5 shuttle vector
-
FLP mediated recombination in E.coli BA5 FRT cells to obtain your desired Ad5 E1/E3 deleted serotype
-
PAC digestion of purified recombinant BAC DNA
-
Virus reconstitution in HEK 293 cells
Licensing Information
If you are interested in this technology, please get in touch with our Business Development and Licensing team at: licensing@sirion-biotech.com or use our contact form.
AdenoBOOST™ Transduction Enhancer
AdenoBOOST is a non-toxic peptide-based solution which is added to the adenovirus particles prior to transduction. The agent specifically bridges the adenovirus surface to the cell membrane, increasing transduction efficiency up to 50-fold. AdenoBOOST is ideal to enhance gene expression in cell types lacking the primary adenovirus CAR-receptor.
Benefits of using AdenoBOOST
-
20 to 50-fold enhanced expression
-
Applicable to almost all cell types without species limitations
-
Reduces the necessary amount of adenovirus particles for any application
-
No cell toxicity
